Metabolomic characterization of frailty identifies subtype-specific management strategies
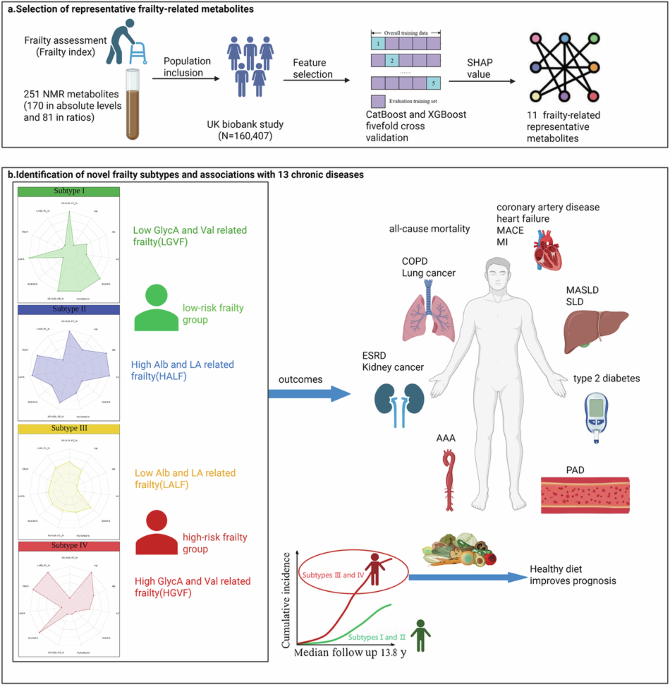
This study identified 11 metabolic features associated with frailty from approximately 160,000 individuals in the UK Biobank using 251 NMR biomarkers and classified four novel frailty subtypes through cluster analysis. Each subtype displayed distinct metabolic and clinical characteristics. Subtypes I and II had a lower risk of chronic diseases compared to subtypes III and IV. Consequently, subtypes I and II were grouped into the low-risk frailty group, while subtypes III and IV were grouped into the high-risk frailty group. Furthermore, our findings demonstrated that high adherence to a healthy diet significantly reduced the risk of chronic diseases in the high-risk frailty group, offering insights to inform personalized clinical decision-making.
Previous studies have applied various methods to categorize frail populations. For instance, research involving approximately 6000 participants identified clustering patterns of multidimensional health issues in older adults, resulting in four subtypes of geriatric frailty26. Another study, conducted in an Asian cohort, explored the relationship between frailty and intrinsic capacity, identifying subgroups with distinct outcomes over a year27. Linzy et al. used a data-driven approach to identify three frailty subtypes—NCF, MTF, and RTF—each exhibiting varying degrees and rates of neurocognitive decline, with MTF showing the steepest trajectory28. Similarly, Okoye et al. identified four clusters of patients with heart failure based on frailty, comorbidities, and B-type natriuretic peptide levels29. In contrast to these studies, our research is the first to conduct a cluster analysis on a large UK-based cohort spanning all ages. We identified subtypes with unique metabolic profiles and examined their associations with 13 chronic diseases and all-cause mortality, contributing to the advancement of personalized medicine.
Among the 11 differential metabolites, GlycA emerged as the most significant. The association between GlycA and frailty can be mechanistically explained through its link to inflammatory pathways, particularly the interleukin-6 (IL-6) signaling axis, which is a known driver of muscle atrophy—a hallmark of frailty. GlycA, a composite biomarker of acute-phase glycoproteins, is associated with various inflammatory markers, including IL-630. The IL-6 pathway is critical in regulating muscle metabolism, where chronic elevation can drive muscle wasting. In experimental models, such as in mice with colon cancer, elevated IL-6 levels have been observed, and IL-6 inhibition was shown to prevent cancer-induced muscle mass loss31. IL-6 exerts its effects by binding to the glycoprotein 130 (GP130) receptor, activating Janus kinases (JAKs) and the signal transducer and activator of transcription 3 (STAT3) pathway. This signaling cascade has been linked to muscle atrophy in numerous contexts. Prolonged IL-6 elevation in cultured myotubes and skeletal muscle cells can lead to increased expression of mitochondrial fission proteins (DRP-1 and FIS-1), which are implicated in muscle atrophy and cellular stress responses31. Thus, elevated GlycA levels in our high-risk subtypes (particularly HGVF) may reflect activation of this IL-6-mediated proteolytic pathway, contributing to the frailty phenotype and its associated adverse outcomes.
Linoleic acid (LA), a polyunsaturated fatty acid (PUFA), showed high SHAP values. Mendelian randomization studies suggest that elevated PUFA levels may prevent frailty32, possibly owing to LA’s antioxidant and anti-inflammatory properties33,34. LA’s influence on metabolic syndrome and related diseases may partly explain the improved prognosis observed in frail individuals with high LA concentrations. Subtype II, characterized by high Alb levels, aligns with research highlighting Alb’s mediating role in frailty and in-hospital mortality among patients with COPD, possibly via inflammatory mechanisms35,36. Conversely, subtype III, with low Alb levels, exhibited a poorer prognosis. This finding reinforces the association between frailty and hypoalbuminaemia, as demonstrated in older adults as well as surgical and hospitalized patients36. Subtype IV was distinguished by elevated Val levels, which may negatively affect frailty by inhibiting muscle synthesis—a hallmark of frailty linked to poor outcomes37,38,39. Restricting Val intake has shown the potential to improve frailty conditions in preclinical models38. It is worth noting that the metabolic profiles defining our frailty subtypes are not formed in isolation but are profoundly influenced by a constellation of behavioral, social, and clinical factors. Socioeconomic status (SES) is a fundamental determinant of health, shaping dietary patterns, access to nutrient-rich foods, and exposure to chronic stress, all of which can directly modulate systemic inflammation (e.g., GlycA levels) and fatty acid metabolism40,41. Consequently, the adverse metabolic signatures observed in our high-risk subtypes (III and IV) may be partially driven by socioeconomic disparities. Furthermore, medication use represents a critical, often necessary, confounder in metabolic studies. For instance, statins drastically alter cholesterol and lipoprotein metabolism42, while metformin and anti-inflammatory drugs can influence insulin sensitivity and inflammatory pathways43. The distinct metabolite levels we observed (e.g., in LA/FA, GlycA) could therefore reflect both the underlying pathophysiology of frailty and the metabolic effects of treatments for its associated comorbidities. Lastly, physical activity is a powerful modulator of the metabolome, influencing energy substrate utilization, insulin sensitivity, and inflammation44. Sedentary behavior, often more prevalent in frail individuals, can lead to ectopic fat accumulation and reduced mitochondrial function, thereby contributing to the pro-inflammatory characteristic of our high-risk subtypes. In summary, while we identified distinct metabolite-driven frailty subtypes, their manifestation is likely orchestrated by a complex interplay between biology, behavior (diet and exercise), social determinants, and clinical management.
Given the limitations of the frailty phenotype and FI45, integrating novel frailty subtypes based on metabolomics and clinical data into routine practice could enhance dynamic monitoring and stratified management of frailty. For the high-risk frailty group, promoting adherence to healthy dietary programs could actively reduce the risk of chronic conditions, such as coronary artery disease, heart failure, MI, MACE, MASLD, SLD, ESRD, COPD, PAD, AAA, lung cancer, kidney cancer, and all-cause mortality. This comprehensive strategy may alleviate the burden of frailty and improve clinical outcomes. In detail, first, for individuals identified as high-risk frailty (Subtypes III and IV), clinicians should prioritize them for intervention. The metabolic characteristics of these patients, such as systemic inflammation (high GlycA), amino acid metabolism dysregulation (high Val), and fatty acid composition imbalance (low LA/FA, high MUFA/FA), provide clear therapeutic targets for intervention. Second, personalized management plans should be developed for this high-risk group. Our research suggests that active lifestyle interventions, particularly nutritional therapies, are most beneficial for them. Based on the results of this study, we propose the following specific dietary recommendations: For Subtype III (LALF: low albumin, low linoleic acid): Encourage the intake of high-quality protein (to boost serum albumin levels) and foods rich in Omega-6 PUFA (such as linoleic acid) (e.g., soybean oil, sunflower oil, nuts) to improve the fatty acid profile. For Subtype IV (HGVF: high GlycA, high valine): The intervention should focus on anti-inflammatory diets (e.g., consuming more Omega-3 rich fish, reducing processed foods and saturated fats) and consider limiting foods high in branched-chain amino acids (BCAAs) (e.g., certain red meats and dairy products) to reduce inflammation and regulate amino acid metabolism.
The limitations of this study should be considered when interpreting the results. First, the UK Biobank cohort predominantly consists of individuals of European descent, with approximately 95% of participants identified as White, as detailed in Table 1. This European bias limits the generalizability of the findings to other racial or ethnic groups. Therefore, caution is warranted when applying these results to more diverse populations, and further validation in Asian and African cohorts is essential to assess the robustness and applicability of these findings across different genetic and environmental contexts. Second, although the NMR metabolomics platform by Nightingale Health offers a comprehensive and standardized metabolite assessment, it does not capture the full blood metabolome and includes a limited range of metabolites. Additionally, some biomarkers lack disease specificity, necessitating further exploration of their links to frailty. Third, randomized controlled trials are essential to validate the clinical utility and impact of these newly defined frailty subtypes. Future research should focus on evaluating their effectiveness and feasibility across different medical settings and populations, considering factors such as cost-effectiveness, resource allocation, and clinical acceptance. Finally, we acknowledge that the absence of spatial transcriptomics and single-cell RNA sequencing data limits our mechanistic understanding of frailty subtypes and chronic diseases. These techniques are crucial for exploring cellular heterogeneity and intercellular interactions. Future research should focus on validating mechanisms such as liver-muscle crosstalk, using these advanced methods to provide deeper insights and identify potential therapeutic targets.
In summary, our study elucidates metabolites associated with frailty, highlights the potential of novel frailty subtypes in managing frail individuals with unhealthy metabolism, and underscores the role of a healthy diet in mitigating the risk of chronic diseases and promoting health.
link






